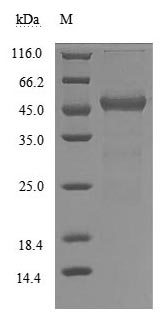
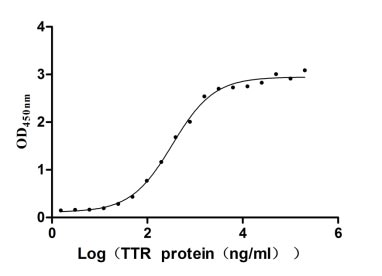
The human transthyretin (TTR) protein (21-147aa) encoding gene and the C-terminal FC-Myc tag gene are respectively inserted into a plasmid vector to form the recombinant plasmid, which is introduced into mammalian cells. Mammalian cells that have successfully taken up the recombinant plasmid can be selected based on their ability to survive in the presence of a specific antibiotic. The recombinant plasmid-containing mammalian cells are cultured under conditions that promote the expression of the interested gene. After expression, the recombinant human TTR protein is isolated and purified from the cell lysate through affinity purification. The resulting recombinant human TTR protein is resolved by denaturing SDS-PAGE, allowing its purity to be estimated, greater than 90%. This protein has been validated as active. In a functional ELISA, this recombinant TTR protein binds to the immobilized RBP4 (CSB-MP019483HU), with the EC50 of 284.7-391.7 ng/ml. Its endotoxin content is less than 1.0 EU/ug as determined by LAL method.
Transthyretin (TTR), also called prealbumin, is a protein made mainly in the liver and brain. It's like a carrier, helping to move important substances like thyroxine and retinol-binding protein in the body's fluids [1] [2] [3] [4] [5]. TTR does a lot: it helps protect the brain, carries thyroid hormones, and supports the growth of nerve cells [6] [7] [8]. It even helps shield against some harmful effects of Alzheimer's disease [6]. But sometimes, TTR can misfold and create amyloid fibrils, causing diseases [9]. Also, it's crucial for carrying vitamin A and can show if someone isn't getting enough nutrients [10] [11].
References:
[1] J. Hamilton and M. Benson, "Transthyretin: a review from a structural perspective", Cellular and Molecular Life Sciences, vol. 58, no. 10, p. 1491-1521, 2001. https://doi.org/10.1007/pl00000791
[2] T. Gião, J. Saavedra, E. Cotrina, J. Quintana, J. Llop, G. Arsequellet al., "Undiscovered roles for transthyretin: from a transporter protein to a new therapeutic target for alzheimer’s disease", International Journal of Molecular Sciences, vol. 21, no. 6, p. 2075, 2020. https://doi.org/10.3390/ijms21062075
[3] M. Liz, T. Coelho, V. Bellotti, M. Fernandez-Arias, P. Mallaina, & L. Obici, "A narrative review of the role of transthyretin in health and disease", Neurology and Therapy, vol. 9, no. 2, p. 395-402, 2020. https://doi.org/10.1007/s40120-020-00217-0
[4] J. Si and J. Kim, "Transthyretin misfolding, a fatal structural pathogenesis mechanism", International Journal of Molecular Sciences, vol. 22, no. 9, p. 4429, 2021. https://doi.org/10.3390/ijms22094429
[5] K. Kusaka, T. Yokoyama, T. Yamada, N. Yano, I. Tanaka, & M. Mizuguchi, "Neutron diffraction experiment with the y116s variant of transthyretin using ibix at j-parc: application of a new integration method", Acta Crystallographica Section D Structural Biology, vol. 76, no. 11, p. 1050-1056, 2020. https://doi.org/10.1107/s2059798320012498
[6] J. Buxbaum, Z. Ye, N. Reixach, L. Friske, C. Levy, P. Daset al., "Transthyretin protects alzheimer's mice from the behavioral and biochemical effects of aβ toxicity", Proceedings of the National Academy of Sciences, vol. 105, no. 7, p. 2681-2686, 2008. https://doi.org/10.1073/pnas.0712197105
[7] J. Gomes, R. Nogueira, M. Vieira, S. Santos, J. Ferraz-Nogueira, J. Relvaset al., "Transthyretin provides trophic support via megalin by promoting neurite outgrowth and neuroprotection in cerebral ischemia", Cell Death and Differentiation, vol. 23, no. 11, p. 1749-1764, 2016. https://doi.org/10.1038/cdd.2016.64
[8] T. Gião, J. Saavedra, J. Vieira, M. Pinto, G. Arsequell, & I. Cardoso, "Neuroprotection in early stages of alzheimer’s disease is promoted by transthyretin angiogenic properties", Alzheimer S Research & Therapy, vol. 13, no. 1, 2021. https://doi.org/10.1186/s13195-021-00883-8
[9] M. Greene, E. Klimtchuk, D. Seldin, J. Berk, & L. Connors, "Cooperative stabilization of transthyretin by clusterin and diflunisal", Biochemistry, vol. 54, no. 2, p. 268-278, 2014. https://doi.org/10.1021/bi5011249
[10] A. Bennekum, S. Wei, M. Gamble, S. Vogel, R. Piantedosi, M. Gottesmanet al., "Biochemical basis for depressed serum retinol levels in transthyretin-deficient mice", Journal of Biological Chemistry, vol. 276, no. 2, p. 1107-1113, 2001. https://doi.org/10.1074/jbc.m008091200
[11] A. Henze, S. Rohn, B. Gericke, J. Raila, & F. Schweigert, "Structural modifications of serum transthyretin in rats during protein‐energy malnutrition", Rapid Communications in Mass Spectrometry, vol. 22, no. 20, p. 3270-3274, 2008. https://doi.org/10.1002/rcm.3728